Document Type : Review Article
Author
Agricultural Chemistry Department, Faculty of Agriculture, Mansoura University, Mansoura, Egypt
Abstract
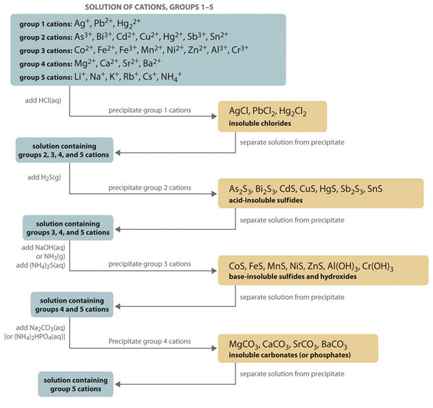
Recent investigation presents exciting, simple and manual procedures to separate and detect different salt radicals of both cations and anions qualitatively and quantitatively. Salt defined as a chemical compound which produce from the reaction of acid with base. So, Acid definition, it is a chemical compound that contains reactable hydrogen (meaning that any positive ion or metal can replace all or part of it) reacts with oxides or hydroxides and gives salt and water. Furthermore, the base is defined as a substance that reacts with acids to produce salt and water only. Basic radicals (Cations) is often a metal, which is the part of the salt shared by the base or alkali in its formation. Basic radicals carry positive charges when ionized and separated from the original salt in water and are called cations and are attracted to the cathode. Acid radicals (Anions) is the part of the salt that is already in the acid from which it is derived or is the part of the salt that the acid shares. Acid radical carries negative charges when ionized and separated from the original salt in water and are called anions and are attracted to the anode. Descriptive analysis is complete when it verifies the presence of basic and acidic radicals in the material to be examined, and often suffices without specifying the truth of the salts formed.
Graphical Abstract
Keywords
Main Subjects
|
HOW TO CITE THIS ARTICLE
Ayman Y. El-Khateeb, Metal Ions of Cations and Anions Separation and Detection Approach, Prog. Chem. Biochem. Res.2020, 3(3), 202-220
DOI: 10.33945/SAMI/PCBR.2020.3.3
|