Document Type : Original Research Article
Authors
Department of Chemistry, Egerton University, P.O Box 536 -20115, Egerton, Kenya
Abstract
Clean water supply and infrastructure is one of the greatest challenges of the 21st century as a result of persistent and emerging water pollutants. The purpose of this study was to determine the concentration profiles of furan and phenol-based contaminants in the boreholes located in the Kerio Valley water basin. The notation KV indicates that the sample was collected in Kerio Valley borehole. The water samples from the boreholes were extracted using a solid phase extraction procedure and characterized by a gas chromatograph interfaced with a mass-selective detector. Based on the findings of this study, 3-methyl-2,3-dihydro-1-benzofuran was significantly present in all the boreholes sampled with a concentration of 9.390±1.12, 0.23±0.02, 0.213±0.05, and 0.070±0.28 ppm in KV1, KV2, KV3, and KV4, respectively. The other major contaminants in the borehole water included 2-furanmethanol and 2-methyltetrahydro-2-furanol. The most toxic phenol, 2,6-dichlorophenol, was found in KV4 with a concentration of 0.06±0.01 ppm. According to the findings of this study, the majority of the borehole water in the Kerio Valley basin is contaminated with furans and phenolic toxins and may not be safe for human consumption. To ensure that residents have access to clean water, it is necessary to develop a policy framework for the evaluation and monitoring of water quality in the area and to suggest remediation strategies.
Graphical Abstract
Highlights
* Phenols and furans are major contaminants in the groundwater of the Kerio Valley basin
* Samples from borehole KV4 had elevated 2,6-dichlorophenol concentration
* Most furan-based contaminants had levels which were above the WHO acceptable limits
* Hydrocarbon exploratory well drilling could be responsible for the observed furans and phenols in borehole water
* Most of the borehole water assessed in this study were highly contaminated and unsafe for domestic use
Keywords
Main Subjects
Introduction
Because of persistent and emerging water pollutants, clean water supply has become one of the greatest challenges witnessed in the 21st century. Therefore, groundwater has been considered the greatest source of potable water globally and the most frequently used for public water supply. This is because groundwater is significantly harder to pollute than surface water although it is more difficult to clean when it is polluted [1]. It is essential to note that water supply issues are of utmost concern globally, especially in situations of water scarcity. Since many aspects of life depend heavily on water, it is important to keep the water clean to safeguard the environment, ecosystems, and people [2]. This study focuses on evaluating the hazardous furan and phenol-based contaminants that compromise water quality in the Kerio Valley water basin. Of significant importance are Tetrahydrofuran, 2-furanmethanol, 2-methyltetrahydro-2-furanol, and bis-phenol, which are well-established toxins and precursors to cancer in water systems.
The Kerio Valley basin in the Kenyan Rift system receives moderate rainfall each year, with the large amount draining into rivers via surface runoff and feeder streams. Consequently, boreholes in the Kerio Valley basin provide abundance and reliable water sources for households and agricultural purposes. Nonetheless, the safety of these groundwater sources has never been investigated, despite concerns about possible contamination from mining activities, and exploration of hydrocarbons taking place in the area.
During the hydrocarbon exploration, hydraulic fracturing technology is employed where a number of harmful chemicals are used in the drilling process, which may contaminate groundwater. As a result, the likelihood of detecting organic contaminants such as furan and phenols in the aquifer system is significantly high. Furthermore, the waste water retrieved from the extraction process ends as in groundwater, resulting in major contamination and modifying the water's quality. Most waste water is held in open pits, which allow organic compounds to seep into the groundwater. Therefore, improper waste water treatment and disposal can cause organic pollution of neighboring water bodies such as rivers and streams.
There are two sources of groundwater pollution globally; either through human activities, or it can occur naturally. Numerous human activities, including farming, industry, and mining, can have a negative impact on groundwater quality [3]. Some human activities that can cause water contamination include hydraulic fracturing during the exploration of hydrocarbons and mining of minerals of economic value – an activity that has been going on in the Kerio Valley water basin for more than five decades. Hydraulic fracturing, used in conjunction with deep horizontal drilling, enables the extraction of natural gas and crude oil from so-called unconventional reservoirs, where they are trapped in tiny rock pores rather than flowing naturally [4]. Fracturing fluid is injected into the borehole under high pressure during this operation. The fluid then induces cracks in the rock via penetrating pipe’s perforations in the horizontal borehole. Proppants, or beads with a diameter of approximately 1 mm, such as sand or ceramic, are employed into the drills with other fracturing fluid. Their purpose is to ''prop'' the pores open when the liquid medium is withdrawn after pressure release. Water is often employed as the liquid media [5]. Fracturing fluids contain chemical additives, which are often added to the water on-site at the drilling location to maintain the proppants suspended in the liquid phase as they travel down to the horizontal pipe until they are deposited in the fissures [6].
In earlier research, the influence of petroleum refinery wastewater on the quality of water in Nigeria's Niger Delta was examined. The findings revealed that waters mixed with the discharged effluent had negative effects on aquatic life [7]. Furthermore, long-term exposure to these harmful hydrocarbons and compounds, such as phenols, can be carcinogenic and result in a variety of serious health problems in humans, including infections of the lungs, liver, kidneys, and vascular systems [8]. Because they are soluble in water and persistent, these contaminants have the potential to enter groundwater. Phenolic chemicals are among the most worrisome persistent pollutants and are mostly produced by the catalytic cracking and fractionation of crude oil in petroleum refineries. In addition, the desalted effluent, waste water from extraction sites, the neutralized wasted caustic waste streams, and tank water are the other sources of total phenolic compounds in the petrochemical effluents. Numerous phenolic chemicals are reportedly released into the environment every year. The normal level of phenols in the waste streams that are discharged might vary, depending on the industrial source of the effluent [9] . This is despite the fact that the release of untreated effluents containing phenol pollutants into the environment, even at low concentrations, can result in the threat to aquatic life and ecosystem harmony as well as the contamination of soil, groundwater, and other surfaces water [10].
The main source of phenolic compound water contamination from an agricultural source is the use of pesticides, insecticides, and herbicides [11]. The detection of phenol, several chlorophenols, including 2,4-dichlorophenol, among others in the aquatic environment has been linked to the degradation of some of these pesticides. These herbicides include 2,4-dichlorophenoxyacetic acid, 4-chloro-2-methylphenoxyacetic acid, and 2,4,5-richlorophenoxyacetic acid. Pentachlorophenol, another pesticide that is widely used in the agricultural sector, eventually breaks down into other chlorophenols with fewer chlorine substituents [12].
Another source of phenolic compounds entering groundwater is through influents and effluents from municipal waste treatment facilities, domestic waste disposals, and leachates from municipal solid waste landfills [13]. From previous studies, cresols have been found in leachates from municipal waste disposal sites and are believed to result from the byproducts of incineration [14]. Similar to 2,4,6-trichlorophenol, 4-tertrabutylphenol, and bisphenol A, which were all observed in leachates and are thought to originate from fly ash, whereas 4-tert-octylphenol was found in landfill leachates and was thought to originate from combustion events [15]. Municipal garbage disposal sites have been shown to include some chlorophenols, 4-nonylphenol, and phenol [16]. As a result, the discharge of untreated leachates from landfills, the discharge of incineration byproducts such as solid fly ash, and the discharge of incombustible materials into neighboring water bodies all contribute to the pollution of the environment.
China [17], Brazil [18], Egypt [19], South Africa [20], India [21], and Malaysia [22] have all published findings about the presence of these priority pollutants in their water bodies. Only a few nations, most notably those in North America (the USA and Canada) and Europe have successfully dealt with phenolic contamination in their water supplies. Because of this, there have not been many reports of these contaminants in their water bodies over the past ten years. On the other hand, a few countries in Asia and Africa, such as China, India, South Africa, and Egypt, have significant concentrations of phenolic compounds, which may be a sign of their countries' expanding industrialization. In addition, a number of developed countries have established permissible concentration limits for the presence of these phenolic compounds in their water, particularly drinking water. For instance, countries in Europe set the maximum level of chlorophenols in drinking water at 0.005 g L -1 [23] while Canada at 0.005 g L -1 [24]. Despite the fact that these phenolic compounds are widely used in industries and particularly for agricultural purposes in Africa, there is a lack of information on their presence in water bodies and drinking water sources within the continent. Consequently, no limit has been established for these priority pollutants in aquatic environments in Kenya and other African countries.
It is reasonable to assume that leachates from urban waste treatment facilities are not the sole source of phenols in the groundwater of the Kerio Valley. Furthermore, effluents from the mining and oil exploration industries, as well as agricultural practices, are also thought to be significant contributors. Oral exposure to phenol is believed to be very hazardous to humans [25]. Chronically exposed humans have reported symptoms such as anorexia, weight loss, diarrhea, vertigo, salivation, dark urine color, and effects on the blood and liver. Animals have had muscular spasms, trouble walking, and even death after ingesting water with incredibly high phenol concentrations. The phenols are definitely cancer-causing to people [26]. The quality of this drinking water from boreholes in Kenya and especially the Kerio Valley, with respect to phenolic chemicals, is largely unidentified since the water is not pre-treated before consumption.
Furan has been identified as a potential groundwater contaminant due to its widespread use and persistence in the environment. Moreover, furan can enter the groundwater through industrial discharges, spills, and leaks from storage tanks [27]. Once in the groundwater, furan can travel great distances and contaminate large volumes of water. It is also known to persist in the environment, which means that it can remain in the groundwater for a long time [28]. Consequently, exposure to furan-contaminated groundwater can pose a risk to human health. Furan has been classified as a potential human carcinogen, which suggests that it may result in cancer. Furan has been associated to long-term liver, kidney, and lung damage as well as reproductive system harm [29].
Furan-contaminated groundwater remediation can be a challenging and expensive operation. Air stripping, activated carbon adsorption and chemical oxidation are prevalent techniques. These techniques work well to remove furan from groundwater but can be expensive and time-consuming [30]. Furan-containing products need to be handled and disposed of carefully to avoid groundwater contamination. Furan-using industries should have adequate spill containment and storage procedures in place to avoid unintentional discharges. Regular monitoring of groundwater near areas where furan is used or stored can also help identify and prevent contamination [31]. The existence of furans in groundwater and their possible consequences on human health and the environment are the subject of numerous ongoing studies. For instance, Zhang et al. (2020) researched the “Furan occurrence in groundwater from a contaminated site and its potential health risk”. This study investigated the occurrence of furans in groundwater samples collected from a contaminated site in China. The authors found that furan concentrations exceeded the drinking water standard in some samples and calculated the potential health risk to humans through ingestion and dermal exposure. Moreover, Faccio et al. (2019) investigated the “Furan contamination of groundwater in an industrial area in Italy”. This study assessed the levels of furans in groundwater samples collected from an industrial area in Italy. The authors found that furan concentrations were higher in samples from the contaminated site compared to a control site and suggest that furan contamination may be linked to industrial activities in the area.
Environmental scientists typically concentrate on the fate, transport, and impact of petroleum hydrocarbons when assessing the environmental impact of oil spills and drilling. The environmental effects of oil spills into groundwater are thought to be reduced by several weathering such as biodegradation, photoxidation, and physical weathering [32]. Hydrocarbons can, however, be transformed into compounds that may be important for the environment through processes like biodegradation and photochemistry. When evaluating the risk of an oil spill, these transformation products are often overlooked [33]. It is imperative to know that oxygenated hydrocarbons are prevalent for longer periods of time and hence considered as persistent in the environment and, therefore, increase the likelihood of their bioaccumulation in an ecosystem as well as the human body [34]. On oxygenated hydrocarbon, there are limited statistics readily available currently on persistence, bioaccumulation, and toxicity (PBT). Therefore, in light of these regulatory requirements, it is crucial to screen the representative chemical families of oxygenated hydrocarbons. Numerous furan and furan-like chemicals have undergone in vivo and in vitro tests to determine the extent of pollution in water systems. Studies on chronic exposure in mammals have shown that furans are linked to poor reproductive results, birth abnormalities, hepatotoxicity, immunosuppression, and carcinogenicity. Mice that consumed furan their entire lives got thyroid and liver cancer. Rats that were similarly exposed got liver, lung, tongue, hard palate, and nose cancers [35].
The detection of furan and phenol-based contaminants at remarkably high concentrations in most of the sampled boreholes is a clear indication that the Kerio Valley groundwater system is not safe for both domestic and commercial applications. However, periodic water quality monitoring of the Kerio Valley water regime is essential to inform both environmental and public health concerns on water quality and safety.
Experimental
Analytical grade (purity ≥99%) reagents were employed in this study. Methanol, hexane, DCM and pure stock furan-D4 solution were purchased from Sigma Aldrich, Inc., St. Louis, Missouri, USA, through its subsidiary, Kobian, Kenya and used without further treatment. A Whatman no.1 and 2 filter paper, C18 cartridge, separating funnel and 2 mL amber vials were purchased from Sigma Aldrich, Inc., St. Louis, Missouri, USA, through its subsidiary, Kobian, Kenya. A rotor vapor, mantle, and Agilent 6890 Gas Chromatograph hyphenated to a mass selective detector (MSD) 5890 series were available in the University laboratory.
Groundwater samples were collected from four boreholes namely; KV1, KV2, KV5, and KV13, along Kerio Valley water basin using sterilized plastic bottle containers, and transported to the laboratory for further treatment and analysis. Whatman no. 1 filter paper was used to filter the sample to eliminate the tiny particles. A binary mixture of 100 mL of 1:1 n-hexane and methanol was added to a 500 mL separating funnel after 300 mL of water sample. After shaking the mixture for 10 minutes to achieve homogeneity, it was left to stand for 30 minutes. Heat was provided to the mantle from a source that operated between 60 and 80 °C. This low-temperature range is advised to prevent the thermal disintegration of molecular components. 500 mL of water from each sampling location was cleaned with a C18 cartridge and filtered using Whatman no. 2 filter paper. A solid-phase extraction approach was used to filter and purify the extract. The extracts were pre-concentrated using a rotor vapor before being sampled into 2 mL amber vials for GC-MS analysis. 20µL of pure stock furan-D4 solution was diluted to 10 mL in methanol to result in a solution of 2µg/mL, which was evaluated for quality assurance/quality control. Under instrumental circumstances that have been previously designed [36, 37], 2 L of the solution combination was injected into the GC-MS.
To guarantee the repeatability of the results, three replicate analyses were carried out. The organic phase was taken and concentrated using a rotor evaporator to 10 mL while the aqueous phase was drawn and discarded. For the extraction of organics, the same method was used on all water samples. Gas chromatograph-mass spectrometry was used to characterize the extract in three replicates.
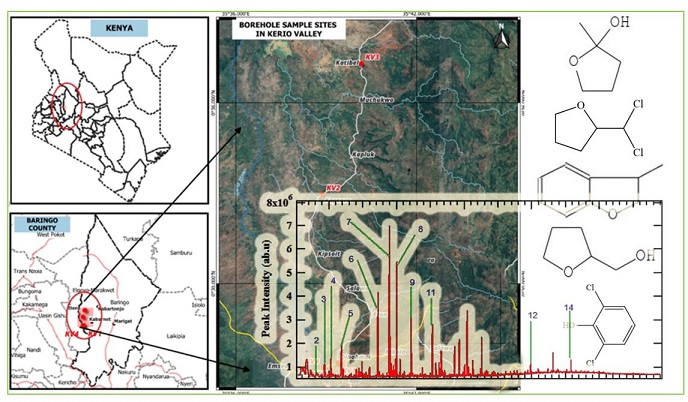
Kerio Valley, the study area (Fig. 1), is located in the Rift Valley part of Baringo County in Kenya. Baringo County is bordered by Turkana County and West Pokot County to the north, Samburu County and Laikipia County to the east, Nakuru County and Kericho County to the south, Uasin Gishu County to the south west and Elkeiyo Marakwet to the west. Kerio Valley lies between 35 ⁰20ʹ0ʺE and 0 ⁰10ʹ0ʺN and covers an area of ~ 50 Km2. It is home to Kenyans whose economic activities include farming, fishing, and fluorspar mining. The source of drinking water is largely boreholes suspected to contain a range of micro-ionic species and hydrocarbons that have not been explored before. Evidently, the quality of water may be affected by rock weathering and waste materials from mining sites that may result in the release of toxic chemicals into both the environment and the aquatic system.
GC-MS characterization of furan and phenol-based contaminants in borehole water
Following the procedure adopted by Bosire, (2016), samples taken from groundwater in the Kerio Valley boreholes were processed through a 10 mL dichloromethane solution (DCM), filtered using Whatman no. 10 filter paper, followed by characterization using Agilent 6890 Gas Chromatograph hyphenated to a mass selective detector (MSD) 5890 series. A filtered sample volume of about 1 L was injected to a GC column (DB-5MS, 30 m, 250 µm, 0.5 µm).
To enable the transformation of organic species into the gas phase prior to MS analysis, the injector port's temperature was set at 200 °C. For programming the temperature, it was heated at a rate of 15 °C/min for 10 minutes, held for 3 minutes at 200 °C, then heated at a rate of 20 °C/min for 5 minutes, held for 10 minutes at 300 °C. An electron impact ionization source of 70 eV was applied in the MS platform. The National Institute of Science and Technology software (NIST, USA) was used to identify the molecular products, and the improved data program built within the Agilent MSD Chemstation provided further confirmation, according to Kibet et al. (2012). Standards of pure compounds were run through the GC-MS under the same circumstances to check that the proper compounds were reported, and it was discovered that the retention periods and peak shapes matched with outstanding precision.
Method validation and recovery test
To ensure repeatability and reproducibility of this study, standards of pure compounds in each category (phenols and furans) were prepared and found to be linear with correlation coefficient,. The spiked borehole samples were used to determine the accuracy of the GC-MS. The relative recovery experiments at various concentrations were assessed and relative recovery values ranged between 88.5% and 105.6% suggesting commendable accuracy. The sensitivity of the GC-MS method was evaluated by computing the limits of detection (LOD) and quantification (LOQ). Ten (10) separate blank solutions were prepared and analyzed. The determination of LOD and LOQ was performed following the protocol advanced by the International Union of Pure and Applied Chemistry (IUPAC) [39] using Equations (1) and (2).
LOD = 3 (1)
LOQ = 10 (2)
Where, s is the slope of the calibration curve for each element and = standard deviation of the measurements of the blank solution.
The calibration curves for each compound were constructed by plotting the peak area of the optimum GC-area counts to the concentration of the standard solution. Next, the least square linear regression analysis was performed to determine the slope, intercept and coefficients.
Boreholes are the primary water sources for agricultural and domestic use in the Kerio Valley basin. Despite concerns about possible pollution from mining companies and the current exploratory wells being conducted in the vicinity in search of hydrocarbons, the safety of these underground water bodies has not previously been assessed.
Based on the findings of this study, furan derivatives such as 2-butyltetrahydrofuran, 2-(dichloromethyl)tetrahydrofuran, 4,5-diethyl-2,3-dimethyl-2,3-dihydrofuran, 2-furanmethanol, 2-methyltetrahydro-2-furanol, 3-methyl-2,3-dihydro-1-benzofuran, 1-(5-ethyltetrahydro-2-furanyl)-3,3-dimethyl-2-butanone, and 5-isopropenyl-3-isopropyl-2,2-dimethyl-2,5-dihydrofuran as reported in Table 1, were present in varied concentrations.
Furan-based contaminants in Kerio Valley borehole water
Based on the findings of this study, 2-(dichloromethyl)tetrahydrofuran was found in borehole water KV1 and KV3 with concentrations of 0.177 ± 0.02 and 0.045 ± 0.01 ppm, respectively lower than the WHO limit of 2.0 ppm allowed in drinking water as shown in Table 2. More importantly, 3-methyl-2,3-dihydro-1-benzofuran was significantly present in all the borehole with concentration of 9.390 ± 1.12, 0.23±0.02, 0.213±0.05, and 0.070±0.28 ppm in decreasing order of KV1, KV2, KV3, and KV4 respectively confirming that it is the major pollutant in the groundwater of Kerio Valley among other furan derivatives.
The representative GC chromatogram of the analyzed organic contaminants reported in KV1 according to the structures presented in Table 1 and Fig 2.
Tetrahydrofuran is a solvent well known to be used in the production of adhesives as used by oil explorers among other significant uses such as production of rubber, resins, plastics, dyes, lacquers, spandex, PVC pipe, and packaging materials [40]. Significantly, tetrahydrofuran spreads readily through soil, but it is also released into the atmosphere and decomposes rapidly. Due to its ease of transition from water to air and potential for biodegradation, tetrahydrofuran has a lower concentration in surface water [41].
Tetrahydrofuran can be found in quite high concentrations in groundwater due to the fact that it is difficult to degrade in groundwater and challenging to move from groundwater to soil and rock thus it has a higher potential to accumulate even beyond the WHO (World Health Organization) permissible limits [42]. Therefore, the difference in concentrations of KV1 and KV4 depends on their distance from exploratory wells injected with the chemical. Nonetheless, this chemical was not detected in KV2. The concentrations of 2-(dichloromethyl)tetrahydrofuran in KV1 is highest compared to that of KV3, as reported in Table 2.
Remarkbaly, 4,5-diethyl-2,3-dimethyl-2,3-dihyd-rofuran was found in KV1 to have concentration of 0.873 ± 0.09 ppm which might have been used during oil exploration in the nearby drills and may have contaminated the groundwater. In addition, 2-furanmethanol and 2-methyltetrahydro-2-furanol were found to be dominant in KV1with 2.64 ± 0.34 ppm and 0.313 ± 0.06 ppm, respectively compared to the WHO standard in drinking water of 0.2 ppm, as reported in Table 2. Notably, these organic compounds are solvents for tanning agents, dyes, and resins in addition to being used in cement, sealants, and as raw materials for furan-based polymers such as urea-formaldehyde [43].
The most significant application is in the production of poly(furfuryl alcohol) resins for sand-based cores and molds in foundries, as well as for the production of plastics, cement, mortars, binders, and adhesives during oil exploration in addition to fungicide and weed killer though it is possibly carcinogenic to human [44]. Generally, the genesis of these organic contaminants in borehole may be attributed to exploratory activities, chemical farming, and poor wastewater management in the study area.
Phenol-based contaminants in Kerio Valley borehole water
To ascertain the relative contributions of drinking water to overall exposure and potential risks to human health, a thorough analysis of bis-phenol compound concentrations was carried out in borehole water from the area of study. From the results, it is clear that KV1 is heavily contaminated with bis-phenol-based compounds such as 3,5-bis(1,1-dimethylethyl)phenol, 2,6-bis(1,1-dimethylethyl)-4-methylphenol, 2,6-bis(1,1-dimethylethyl)-4-ethylphenol, and 2,6-bis(1,1-dimethylpropyl)-4-methylphenol, in the order of decreasing concentration of 5.99, 1.11, 0.69 and 0.134 ppm, respectively, as presented in Table 3.
It is significant to note that 3,5-bis(1,1-dimethylethyl)phenol concentration is twice the WHO limits in drinking water as used by the inhabitants and approximately fifty times more than the concentration reported in KV2. Nonetheless, 2,6-bis(1,1-dimethylethyl)-4-methylphenol and 2,6-bis(1,1-dimethylpropyl)-4-methylphenol concentration in groundwater of KV1 is 1.11 ± 0.07 and 0.134 ± 0.03 ppm, respectively, as provided in Table 3 . This concentration is significantly higher than the WHO allowable limit in drinking water even though 2,6-bis(1,1-dimethylpropyl)-4-methylphenol was lower than the toxic concentration limit in aquatic environments.
3-(1,1-dimethylethyl)-4-methoxylphenol and 2-(1,1-dimethylethyl)-4-methoxyphenol were also dominant in borehole water KV1. The toxicology of these pollutants in humans includes liver and kidney damage, skin discoloration, and eye damage, and may also result in vision impairment, as adversely witnessed in the Kerio Valley region [45]. The concentration of 3-(1,1-dimethylethyl)-4-methoxylphenol and 2-(1,1-dimethylethyl)-4-methoxyphenol were noted to be 9.40 ± 1.20 and 0.09 ± 0.02 ppm, respectively compared to the WHO limit of 2.5 ppm, as shown in Table 3. Nonetheless, 2,6-dichlorophenol and 2-analino-4,6-di-tert-butylphenol emerged in KV4 water sample in concentrations of 0.06 ± 0.01 ppm and 0.103 ± 0.02 ppm, respectively. More importantly, these pollutants are toxic at elevated concentrations in aquatic environments, and thus, discharges from mining industries, as well as continuous chemical methods of farming, need to be inspected more often in order to mitigate challenges posed to the environment [46].
Apart from other pollutants in KV2, 1,4-epoxynaphthalene-1(2H)-methanol posted a higher concentration of 2.804 ppm, which is about nine times more than the WHO limits for oral ingestion, as summarized in Table 3. The pollutants reported in this work may be as a result of poorly managed wastewater and discharges from mining companies within the Kerio Valley region, municipal waste as well as continuous chemical farming taking place in the area.
According to the findings of this investigation, the Kerio Valley water boreholes are contaminated with furan and phenol-based organic pollutants. Although some boreholes near the hydrocarbon exploratory wells were analyzed, it is possible to believe that the majority of borehole water in this region is adversely contaminated by these organic pollutants and may be unfit for human consumption; in addition the borehole water is unsuitable for irrigation. Remarkably, 3-methyl-2,3-dihydro-1-benzofuran and 2-furanmethanol showed much greater quantities than other furan based pollutants in the KV1 borehole, with 9.390 and 2.64 ppm, respectively. The concentration is significantly higher compared to the WHO permissible limit. Moreover, KV1 had significant 3-(1,1-dimethylethyl)-4-methoxyphenol, 3,5-bis(1,1-dimethylethyl)phenol, and 2,6-bis(1,1-dimetylethyl)-4-methylphenol concentration profiles of 9.40, 5.99, and 1.11 ppm, respectively. However, KV4 had an elevated 2,6-dichlorophenol concentration of 0.06 ppm, which is approximately sixty times the WHO recommended levels in drinking water. According to these data, the KV1 and KV4 boreholes are by far the most polluted of all the other boreholes analyzed in this study. When examining the human and environmental impact, it is usually impossible to identify the losses produced by the current exposure. The impacts of potential water pollution must be carefully considered throughout hydrocarbon exploration and any other mining activity.
Surface and groundwater monitoring methods and programs, as well as ambient air monitoring, must be modified by health authorities. Groundwater studies have indicated a high link between reported health consequences and hydraulic fracturing leachates as well as other mining activities taking place beneath the Earth's surface. Since epidemiological studies typically describe human exposure using various distance measures as a surrogate, the challenge for future studies will be use not only measured concentrations of pollutants, but also the kinetics and characterization of organic pollutants during hydraulic fracturing and mining activities beneath the ground surface. For both humans as well as the environment, the use of explosives and fracturing using hazardous organic compounds continue to be detrimental to both public and environmental health.
ORCID
Festus K. Langat
https://orcid.org/0009-0009-9106-9145
Joshua K. Kibet
https://orcid.org/0000-0002-9924-961X
Francis I. Okanga
https://orcid.org/0009-0009-8249-4502
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Declarations
Conflict of interest: The authors have no relevant financial or non-financial interests to disclose.
Ethical approval: Not applicable.
Consent to participate: Not applicable.
Consent for publication: Not applicable
----------------------------------------------------------------------------------------------------------------------------------------------------
OPEN ACCESS
©2024 The author(s). This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit: http://creativecommons.org/licenses/by/4.0/
PUBLISHER NOTE
Sami Publishing Company remains neutral concerning jurisdictional claims in published maps and institutional affiliations.
CURRENT PUBLISHER
Sami Publishing Company
- Sarker B, Keya KN, Mahir FI, Nahiun KM, Shahida S, Khan RA. Surface and ground water pollution: causes and effects of urbanization and industrialization in South Asia. Scientific Review. 2021;7(3):32-41. [Crossref], [Google Scholar], [Publisher]
- Singh J, Yadav P, Pal AK, Mishra V. Water pollutants: Origin and status. Sensors in water pollutants monitoring: Role of material. 2020:5-20. [Crossref], [Google Scholar], [Publisher]
- Burri NM, Weatherl R, Moeck C, Schirmer M. A review of threats to groundwater quality in the anthropocene. Science of the Total Environment. 2019;684:136-54. [Crossref], [Google Scholar], [Publisher]
- Jew AD, Druhan JL, Ihme M, Kovscek AR, Battiato I, Kaszuba JP, et al. Chemical and reactive transport processes associated with hydraulic fracturing of unconventional oil/gas shales. Chemical reviews. 2022;122(9):9198-263. [Crossref], [Google Scholar], [Publisher]
- Zhang K, Zhang G, Li Z, Hou J. Laboratory evaluation of a low pH and low polymer concentration zirconium-CMHPG gel system for hydraulic fracturing. Energy & Fuels. 2019;33(10):9720-35. [Crossref], [Google Scholar], [Publisher]
- Peers De Nieuwburgh C. Characterising organic and inorganic chemical signatures of water associated with shale gas extraction operations. 2020. [Crossref], [Google Scholar],
- Howard IC, Okpara KE, Techato K. Toxicity and Risks Assessment of Polycyclic Aromatic Hydrocarbons in River Bed Sediments of an Artisanal Crude Oil Refining Area in the Niger Delta, Nigeria. Water. 2021;13(22):3295. [Crossref], [Google Scholar], [Publisher]
- Kuppusamy S, Maddela NR, Megharaj M, Venkateswarlu K, Kuppusamy S, Maddela NR, et al. Impact of total petroleum hydrocarbons on human health. Total Petroleum Hydrocarbons: Environmental Fate, Toxicity, and Remediation. 2020:139-65. [Crossref], [Google Scholar], [Publisher]
- Ahmed J, Thakur A, Goyal A. Industrial wastewater and its toxic effects. 2021. [Crossref], [Google Scholar], [Publisher]
- Akpan CO, Bassey SA. The quandary on water pollution in Nigeria’s Niger Delta: an environmental ethical analysis. 2020. [Crossref], [Google Scholar], [Publisher]
- Idowu GA, David TL, Idowu AM. Polycarbonate plastic monomer (bisphenol-A) as emerging contaminant in Nigeria: Levels in selected rivers, sediments, well waters and dumpsites. Marine Pollution Bulletin. 2022;176:113444. [Crossref], [Google Scholar], [Publisher]
- Leung KT, Jiang Z-H, Almzene N, Nandakumar K, Sreekumari K, Trevors JT. Biodegradation and bioremediation of organic pollutants in soil. 3rd ed. Modern soil microbiology. 2019:381-402. [Google Scholar], [Publisher]
- El Fadili H, Ali MB, El Mahi M, Cooray AT. A comprehensive health risk assessment and groundwater quality for irrigation and drinking purposes around municipal solid waste sanitary landfill: A case study in Morocco. Environmental Nanotechnology, Monitoring & Management. 2022;18:100698. [Crossref], [Google Scholar], [Publisher]
- Kumar V, Sharma N, Umesh M, Chakraborty P, Kaur K, Duhan L, et al. Micropollutants characteristics, fate, and sustainable removal technologies for landfill leachate: A technical perspective. Journal of Water Process Engineering. 2023;53:103649. [Crossref], [Google Scholar], [Publisher]
- Kotowska U, Kapelewska J, Sawczuk R. Occurrence, removal, and environmental risk of phthalates in wastewaters, landfill leachates, and groundwater in Poland. Environmental Pollution. 2020;267:115643. [Crossref], [Google Scholar], [Publisher]
- Ateş H, Argun ME. Advanced oxidation of landfill leachate: Removal of micropollutants and identification of by-products. Journal of Hazardous Materials. 2021;413:125326. [Crossref], [Google Scholar], [Publisher]
- Wu Y, Zhong L, Yuan J, Xiang W, Xin X, Liu H, et al. Photocatalytic optical fibers for degradation of organic pollutants in wastewater: a review. Environmental Chemistry Letters. 2021;19:1335-46. [Crossref], [Google Scholar], [Publisher]
- Ramos RL, Moreira VR, Lebron YA, Santos AV, Santos LV, Amaral MC. Phenolic compounds seasonal occurrence and risk assessment in surface and treated waters in Minas Gerais—Brazil. Environmental Pollution. 2021;268:115782. [Crossref], [Google Scholar], [Publisher]
- El-Naggar NA, Moawad MN, Ahmed EF. Toxic phenolic compounds in the Egyptian coastal waters of Alexandria: spatial distribution, source identification, and ecological risk assessment. Water Science. 2022;36(1):32-40. [Crossref], [Google Scholar], [Publisher]
- Yahaya A, Okoh OO, Agunbiade FO, Okoh AI. Occurrence of phenolic derivatives in Buffalo River of Eastern Cape South Africa: exposure risk evaluation. Ecotoxicology and Environmental Safety. 2019;171:887-93. [Crossref], [Google Scholar], [Publisher]
- Othmani A, Magdouli S, Kumar PS, Kapoor A, Chellam PV, Gökkuş Ö. Agricultural waste materials for adsorptive removal of phenols, chromium (VI) and cadmium (II) from wastewater: A review. Environmental Research. 2022;204:111916. [Crossref], [Google Scholar], [Publisher]
- Lim KL. Photocatalytic degradation of Rhodamine B in greywater over ZnO/BiObr/MgFe2O4 Ternary Nanocomposite under sunlight irradiation: performance evaluation, kinetic and phytotoxicity studies: UTAR; 2022. [Google Scholar], [Publisher]
- Saxena G, Chandra R, Bharagava RN. Environmental pollution, toxicity profile and treatment approaches for tannery wastewater and its chemical pollutants. Reviews of Environmental Contamination and Toxicology Volume 240. 2017:31-69. [Crossref], [Google Scholar], [Publisher]
- Wu Y, Xu X, McCarter CP, Zhang N, Ganzoury MA, Waddington JM, et al. Assessing leached TOC, nutrients and phenols from peatland soils after lab-simulated wildfires: Implications to source water protection. Science of The Total Environment. 2022;822:153579. [Crossref], [Google Scholar], [Publisher]
- Chowdhary P, Bharagava RN, Mishra S, Khan N. Role of industries in water scarcity and its adverse effects on environment and human health. Environmental Concerns and Sustainable Development: Volume 1: Air, Water and Energy Resources. 2020:235-56. [Crossref], [Google Scholar], [Publisher]
- Thakur A, Kumar A. Recent advances on rapid detection and remediation of environmental pollutants utilizing nanomaterials-based (bio) sensors. Science of The Total Environment. 2022:155219. [Crossref], [Google Scholar], [Publisher]
- Encarnação T, Pais AA, Campos MG, Burrows HD. Endocrine disrupting chemicals: Impact on human health, wildlife and the environment. Science progress. 2019;102(1):3-42. [Crossref], [Google Scholar], [Publisher]
- Saxena P, Sarkar A, Hazarika R, Pattanaik A, Yadav OP, Achari G. Exploring persistent organic pollutants (POPs) in the Danube River: An analysis of Polybrominated diphenyl ethers (PBDEs) and Per-and Polyfluorinated Substances (PFAS) using NORMAN Database. 2023. [Crossref],
- Arcenal KJ, Cordero RR, Laluna CR, Marfe D. A Review on the Toxicological Elements of Commercial Pediatric Food and its Long-Term Bodily Effects. Journal homepage: www ijrpr com ISSN.2582:7421. [Crossref], [Google Scholar], [PDF]
- Naha A, Antony S, Nath S, Sharma D, Mishra A, Biju DT, et al. A hypothetical model of multi-layered cost-effective wastewater treatment plant integrating microbial fuel cell and nanofiltration technology: A comprehensive review on wastewater treatment and sustainable remediation. Environmental Pollution. 2023:121274. [Crossref], [Google Scholar], [Publisher]
- Khobragade DS. Health care waste: Avoiding hazards to living and non living environment by efficient management. Fortune Journal of Health Sciences. 2019;2(2):14-29. [Crossref], [Google Scholar], [PDF]
- Kuppusamy S, Maddela NR, Megharaj M, Venkateswarlu K, Kuppusamy S, Maddela NR, et al. Fate of total petroleum hydrocarbons in the environment. Total Petroleum Hydrocarbons: Environmental Fate, Toxicity, and Remediation. 2020:57-77. [Crossref], [Google Scholar], [Publisher]
- Ward CP, Overton EB. How the 2010 Deepwater Horizon spill reshaped our understanding of crude oil photochemical weathering at sea: a past, present, and future perspective. Environmental Science: Processes & Impacts. 2020;22(5):1125-38. [Crossref], [Google Scholar], [Publisher]
- Kirkok SK, Kibet JK, Kinyanjui TK, Okanga FI. A review of persistent organic pollutants: Dioxins, furans, and their associated nitrogenated analogues. SN Applied Sciences. 2020;2:1-20. [Crossref], [Google Scholar], [Publisher]
- Cholico GN. Acute Exposure to TCDD Increases Liver Disease Progression in Mice With Carbon Tetrachloride-Induced Liver Injury. 2019. [Crossref], [Google Scholar], [Publisher]
- Kibet JK, Khachatryan L, Dellinger B. Phenols from pyrolysis and co-pyrolysis of tobacco biomass components. Chemosphere. 2015;138:259-65. [Crossref], [Google Scholar], [Publisher]
- Christine AA, Kibet JK, Kiprop AK, Were ML. The assessment of bore-hole water quality of Kakamega County, Kenya. Applied water science. 2018;8:1-8. [Crossref], [Google Scholar], [Publisher]
- Langat FK, Kibet JK, Okanga FI. Heavy Metal Analysis in the Ground Water of Kerio Valley Sub-Water Basin, Baringo County, Kenya. Environ Sci Arch. 2024;3(1):58-66. [Google Scholar], [Publisher], [Crossref]
- Bosire JOe. Particulate emissions from selected combustion sources and their pathological impacts on the lung tissues of male albino mice: Egerton University; 2016. [Google Scholar], [Publisher]
- Marques AC, Mocanu A, Tomić NZ, Balos S, Stammen E, Lundevall A, et al. Review on adhesives and surface treatments for structural applications: Recent developments on sustainability and implementation for metal and composite substrates. Materials. 2020;13(24):5590. [Crossref], [Google Scholar], [Publisher]
- Yang L, Shi F, Yang J. Experimental studies on hydraulic fracturing in hydrate sediment. Chemistry and technology of fuels and oils. 2020;56:107-14. [Crossref], [Google Scholar], [Publisher]
- Takai S, Shimada T, Takeda S, Koike K. Evaluating the effectiveness of a geostatistical approach with groundwater flow modeling for three-dimensional estimation of a contaminant plume. Journal of Contaminant Hydrology. 2022;251:104097. [Crossref], [Google Scholar], [Publisher]
- Palaniappan SK, Chinnasamy M, Rathanasamy R, Pal SK. Synthetic Binders for Polymer Division. Green Adhesives: Preparation, Properties and Applications. 2020:227-72. [Crossref], [Google Scholar], [Publisher]
- Pizzi A, Ibeh C. Phenol-formaldehyde resins. Handbook of Thermoset Plastics: Elsevier; 2022. p. 13-40. [Crossref], [Google Scholar], [Publisher]
- Program NT. NTP toxicology and carcinogenesis studies of trans-cinnamaldehyde (CAS No. 14371-10-9) in F344/N rats and B6C3F1 mice (feed studies). National Toxicology Program technical report series. 2004(514):1-281. [Crossref], [Google Scholar], [Publisher]
- Pan J, Yao H, Guan W, Ou H, Huo P, Wang X, et al. Selective adsorption of 2, 6-dichlorophenol by surface imprinted polymers using polyaniline/silica gel composites as functional support: equilibrium, kinetics, thermodynamics modeling. Chemical engineering journal. 2011;172(2-3):847-55 [Crossref], [Google Scholar], [Publisher]
Citation: Festus K. Langat, Joshua K. Kibet, Francis I. Okanga. Furan and Phenol-Based Contaminants in the Borehole Water Quality of the Kerio Valley Water Basin, Kenya. Prog. Chem. Biochem. Res., 7(2) (2024) 143-153