Document Type : Original Research Article
Author
Department of Chemical Engineering, University of Bojnord, Bojnord, Iran
Abstract
Gas hydrates could provide a clean and sustainable option to meet global energy demands. Accelerating gas hydrate formation is crucial to exploiting its positive applications, as these ice-like structures can serve as a vast and clean source of energy. In this research, we delve into the influence of sodium dodecyl sulfate (SDS) on the growth rate of double tetra n-butylammonium chloride (TBAC) + methane semiclathrate hydrate. To investigate this, we conducted experiments utilizing a stirred batch cell with a total volume of 169 cm3. The cell's temperature was maintained at 278.15 K, while the initial pressures were set at 6 and 8 MPa. Comparing the results with pure water, SDS showed a significant positive effect on the growth rate of methane hydrate formation. However, when 400 ppm of SDS was introduced, it had a detrimental impact on the average growth rate of the double (TBAC + methane) semiclathrate hydrate within just 50 minutes of the process.
Graphical Abstract
Highlights
- A thermodynamic and kinetic promoter mixture is used for hydrate formation
- The addition of 3 and 5 wt% TBAC increased the average growth rate of hydrate formation
- Using TBAC and SDS together decreased growth rate compared to SDS solution and TBAC solution.
Keywords
Main Subjects
- INTRODUCTION
Methane hydrates, also known as methane clathrates, represent a unique class of crystalline structures in which methane molecules are encaged within cavities formed by molecules of water. These hydrates occur in nature under specific conditions of high-pressure and low-temperature, commonly found in permafrost regions and marine sediments [1]. Gas hydrates have a wide range of positive utilization. They can be used for storage of gases, gas separation, wastewater treatment, juice concentration, energy recovery, gas transportation, water desalination, and CO2 capturing [2-8]. Despite the immense promise methane hydrates hold as a clean energy resource, their commercial viability hinges on understanding and controlling the kinetics of their formation [5, 9-11]. The formation process involves the nucleation of hydrate crystals and their subsequent growth, which is influenced by various factors, including temperature, pressure, guest molecules and the presence of additives [12-14]. A thorough comprehension of the kinetics is essential to develop efficient and cost-effective methods for hydrate production. Moreover, an in-depth study of formation of gas hydrates in the presence of specific additives can shed light on their potential role in enhancing or inhibiting the process, leading to improved exploitation strategies.
Three typical structures can be identified when it comes to gas hydrates, including: Structure sI that is the most common and well-studied gas hydrate structure. It consists of water molecules forming hexagonal cages with one central guest molecule. The cages are arranged in a cubic lattice structure. Structure sII is similar to Structure sI, but has a larger cage size. The water molecules form pentagonal dodecahedral cages, accommodating multiple guest molecules. It is often found at higher pressures and structure sH that has a hexagonal prism structure and consists of two types of cages - large and small. Large cages contain guest molecules, while small cages can be empty. This structure is commonly found in certain clathrates, including gas hydrates [1, 15]. In 1940, Fowler et al. made a groundbreaking discovery of a novel hydrate structure formed by tetra n-butyl ammonium salts. These structures, known as semiclathrates, involve the replacement of certain water lattice sites with guest molecules (anions) [16-21]. Semiclathrate hydrates (SCHs) are a promising phase change material (PCM) for storing and transporting the cold energy, as well as for improving the energy efficiency of cooling processes [21-24].
The phase equilibrium of semiclathrate hydrates has been investigated by numerous researchers in recent years [25-31]. However, despite the understanding of the thermodynamics governing semiclathrate hydrate formation, there has been relatively less emphasis on studying the kinetics of these hydrates [32-34]. The aim of this study is to explore how the growth rate of double (TBAC+ methane) semiclathrate hydrate is affected by SDS. This research provides insight into the differentiated behavior of gas hydrate formation in the SDS presence and its implications for energy-related and other applications. This highlights the need for further research on the role of additives in shaping the properties and behavior of gas hydrates to pave the way for innovative and sustainable energy solutions. The innovation of this study is the targeted study of the effects of SDS on the growth rate of specific gas hydrate structures, investigating both beneficial and detrimental effects, and providing useful insights in the fields of energy, environment, and materials science.
- EXPERIMENTAL
2.1. Materials and Apparatus
The information regarding the chemicals used in this study, including their names, purities, molecular structures, and suppliers, is presented in Table 1. A visual representation of the experimental setup employed to study the kinetics of methane+TBAC semiclathrate hydrate formation is displayed in Fig 1.
The main component of the equipment is a high-pressure cell with an inner volume of 169 cm3 and a maximum operating pressure of 20 MPa. To ensure proper mixing within the cell, a rocking cell stirrer is utilized, which operates at a speed of 10 rpm. The temperature of the cell is carefully regulated using a cooling system that circulates a mixture of 50% ethylene glycol and water as a coolant. Pressure measurements are taken using a 0.01 MPa scale pressure transducer, while temperatures are monitored using a 0.1 K scale thermocouple (PT100).

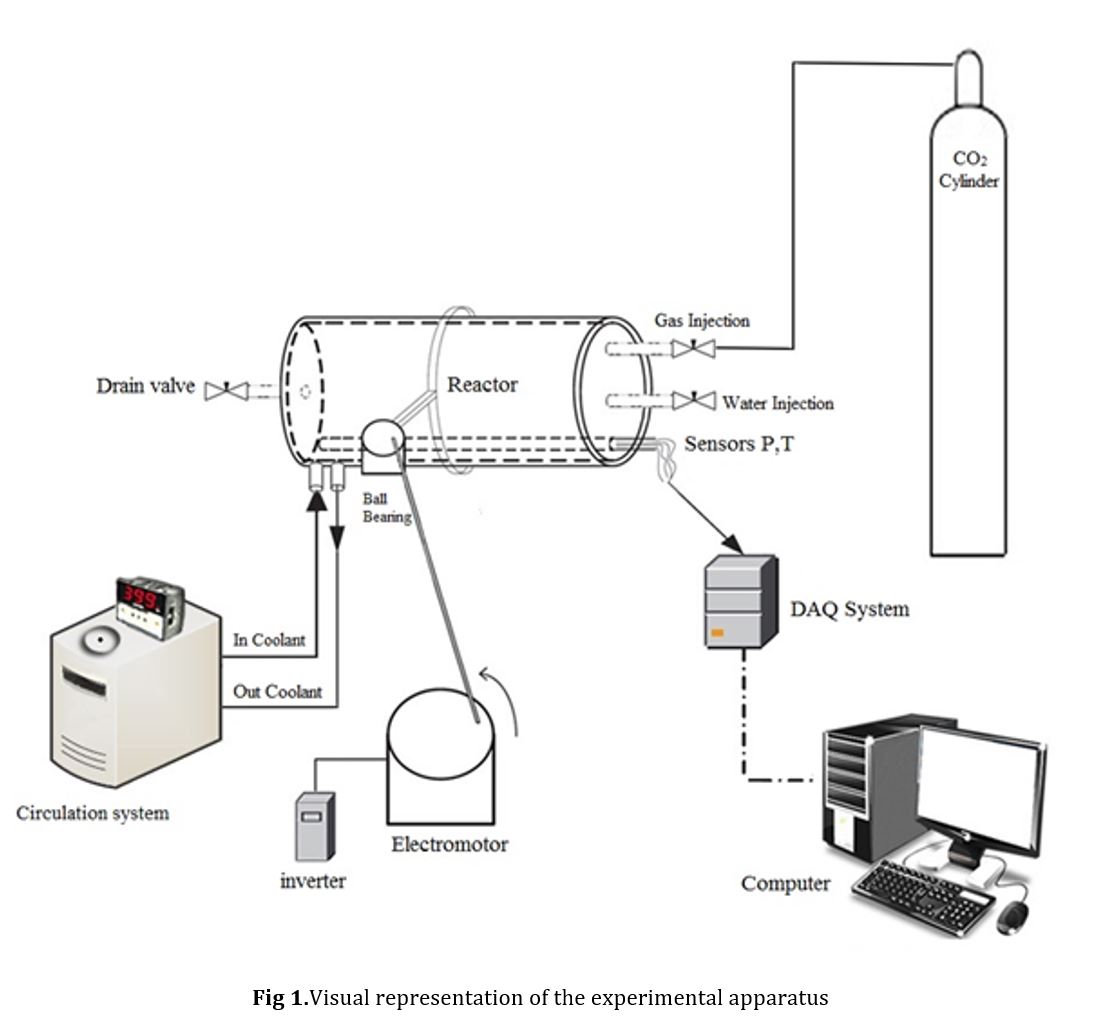
2.2. Procedure
Various solutions of TBAC and SDS were prepared in an aqueous form. Afterwards, a precise amount of the prepared solution was added to the batch crystallizer. The temperature of the crystallizer was regulated to 278.15 K by means of a circulator. Gas was subsequently introduced into the crystallizer until the desired pressure levels were reached. To agitate the solution within the crystallizer, a rocking cell stirrer was activated at a speed of 10 rpm.
- RESULTS AND DISCUSSION
To examine the influence of SDS on the growth rate of (TBAC+methane) semiclathrate hydrate various aqueous solutions were prepared, including: (a) Aqueous solutions containing 3 wt% and 3 wt% TBAC, (b) An aqueous solution with 400 ppm SDS, and (c) A TBAC + SDS mixture. Throughout the experiments, a constant temperature of 278.15 K and pressure of 6 MPa and 8 MPa were maintained within the cell to study the rate of TBAC+methane semiclathrate hydrate formation under these conditions.
The average growth rate (Rav) within the first 50 minutes of the process was evaluated using the following equation which was used to evaluate the average rate of gas uptake (R50) within 50 min of the process. represent the number of moles of methane converted to gas hydrate, the number of moles of feed water used, and the time period, respectively.
Equation (2) was utilized to determine the quantity of methane uptake throughout the 50 min of the process. Where, and , respectively, stand for the methane mole number in the gas phase at time t = 0 and time 50 min, and can be calculated by the following equations. Where, subscripts 0 and 50, respectively, are the reactor conditions at beginning and time 50 min, and Z is evaluated using PR EOS [35]. Table 1 presents the growth rate of TBAC + methane semiclathrate hydrate during the first 50 minutes of the process. Fig 2 and 3 illustrate the influence of TBAC on the growth rate of methane hydrate per mole of feed solution at a constant T = 278.15 K and pressures of 6 MPa and 8 MPa, respectively.


Based on the data presented in Fig 2 and 3, as well as Table 1, it is evident that the inclusion of 3 wt% and 5 wt% of TBAC has not a significant influence on the average growth rate of methane at initial pressure of 6 MPa, while a positive effect can be seen at P0 = 8 MPa.
At P0 = 8 MPa, the growth rate of formed hydrate in water is 0.40 mmol/mol.s, whereas in the TBAC solution (5 wt%), it is 0.70 mmol/mol.s. This indicates that the average growth rate of formed hydrate in the TBAC solution is 75% higher than in pure water under the mentioned thermodynamic conditions. By incorporating TBAC, the equilibrium pressure of methane hydrate formation is reduced, leading to a substantial increase in the driving force behind the process. This significant reduction in pressure moderates the thermodynamic conditions during methane hydrate formation [37-41]. By moderating Thermodynamics conditions, such as reducing the equilibrium pressure through the utilization of TBAC, the growth rate of the process is increased. This is because the reduction in pressure enhances the driving force of the process, leading to more efficient and faster methane uptake into the gas hydrate structure.


In Fig 4 and 5, and as supported by the data in Table 1, a comparison was made between the influence of SDS alone and SDS + TBAC on the growth rate of TBAC+methane semiclathrate hydrate. The investigation revealed that the average growth rate of TBAC+methane semiclathrate hydrate for the solution of SDS+TBAC is lower than that of the SDS solution, consistently observed at 278.15 K and both tested pressures. This negative effect on the growth rate might be attributed to the interaction between the released ions of TBAC and SDS within the aqueous solution. Such interactions could lead to changes in the process, affecting the growth rate of the hydrate crystals.

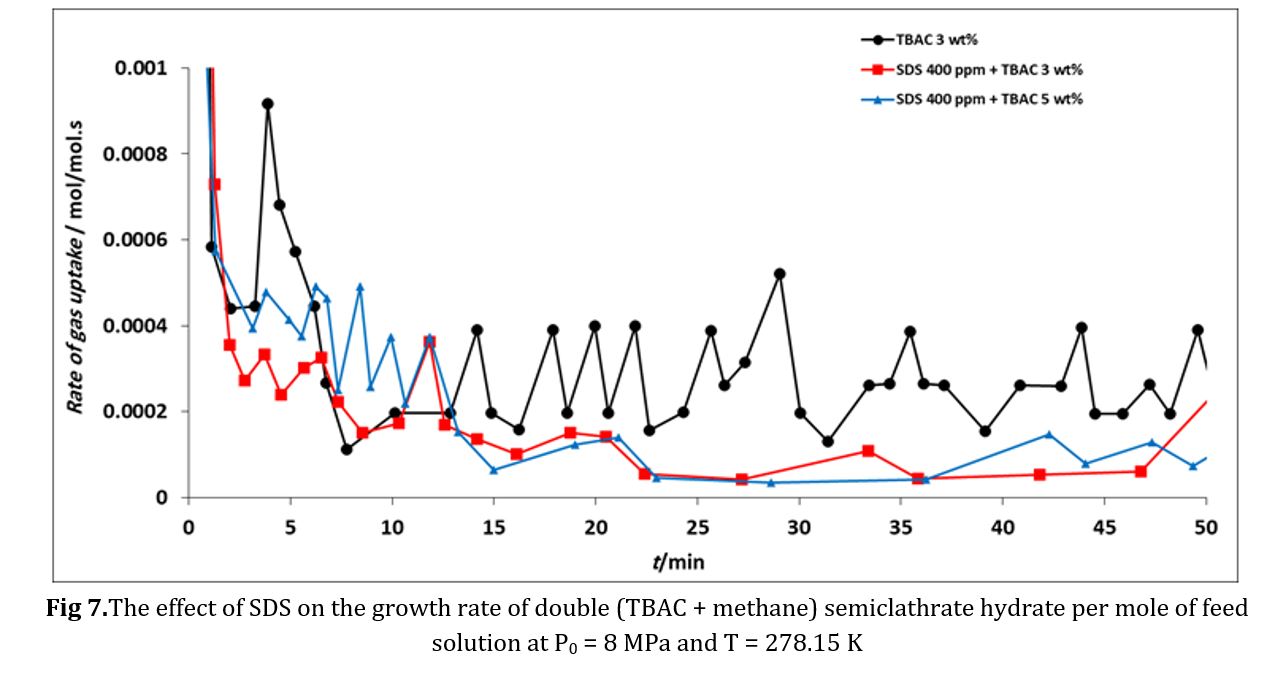

Fig 6 and 7 compare the growth rates of (TBAC + methane) semiclathrate hydrate under the influence of TBAC and SDS+TBAC. The results revealed that in SDS+TBAC solutions, the growth rate of TBAC+methane semiclathrate hydrate is lower than when only TBAC is present.
Fig. 8 illustrates the impact of various concentrations of used additives (SDS and TBAC) on the average growth rate of formed hydrate at 278.15 K and P0 = 6 and 8 MPa. From the figure, it is evident that using SDS (400 ppm) yields the most significant improvement in the rate of gas uptake. However, when SDS and TBAC are present together, the average rate of methane uptake is lower than that observed in the both SDS-only TBAC-only solutions.
- CONCLUSION
The influence of SDS on the average growth rate of (TBAC+methane) semiclathrate hydrate within a 50-minute period was investigated. Experimental results obtained at 278.15 K and P0 = 6 and 8 MPa demonstrated that the addition of 3 wt% and 5 wt% TBAC led to an increase in the average rate of methane uptake. However, when both TBAC and SDS were used simultaneously, their interaction resulted in a negative effect on the rate of methane uptake compared to the use of the aqueous solution of SDS alone.
ORCID
- Mohammadi
https://orcid.org/0000-0002-0623-4815
- Sloan Jr ED, Koh CA. Clathrate hydrates of natural gases, CRC press; 2007 Sep 7.
- Dong H, Wang J, Xie Z, Wang B, Zhang L, Shi Q. Potential applications based on the formation and dissociation of gas hydrates, Renewable and Sustainable Energy Reviews; 2021 Jun 1; 143:110928. https://doi.org/10.1016/j.rser.2021.110928
- Mohammadi A. The roles TBAF and SDS on the kinetics of methane hydrate formation as a cold storage material, Journal of Molecular Liquids; 2020 Jul 1; 309:113175. https://doi.org/10.1016/j.molliq.2020.113175
- Pahlavanzadeh H, Javidani AM, Ganji H, Mohammadi A. Investigation of the Effect of NaCl on the Kinetics of R410a Hydrate Formation in the Presence and Absence of Cyclopentane with Potential Application in Hydrate-based Desalination, Industrial & Engineering Chemistry Research; 2020 Jul 14; 59(31):14115-25. https://doi.org/10.1021/acs.iecr.0c02504
- Farhadian A, Naeiji P, Varfolomeev MA, Peyvandi K, Kiiamov AG. Reconsideration of the micellization theory: Promotion or inhibition of gas hydrate formation for gas storage and flow assurance applications, Chemical Engineering Journal; 2022 Jan 1; 427:131852. https://doi.org/10.1016/j.cej.2021.131852
- Abedi-Farizhendi S, Hosseini M, Iranshahi M, Mohammadi A, Manteghian M, Mohammadi AH. Kinetics of CO2 hydrate formation in coffee aqueous solution: Application in coffee concentration, Journal of Dispersion Science and Technology; 2019 May 29. https://doi.org/10.1080/01932691.2019.1614031
- Abdi-Khanghah M, Adelizadeh M, Naserzadeh Z, Barati H. Methane hydrate formation in the presence of ZnO nanoparticle and SDS: Application to transportation and storage, Journal of Natural Gas Science and Engineering; 2018 Jun 1; 54:120-30. https://doi.org/10.1016/j.jngse.2018.04.005
- Dashti H, Yew LZ, Lou X. Recent advances in gas hydrate-based CO2 capture, Journal of Natural Gas Science and Engineering; 2015 Mar 1; 23:195-207. https://doi.org/10.1016/j.jngse.2015.01.033
- Chirkova YF, Mirzakimov UZ, Semenov ME, Pavelyev RS, Varfolomeev MA. Promising Hydrate Formation Promoters Based on Sodium Sulfosuccinates of Polyols, Energies; 2022 Dec 28; 16(1):359. https://doi.org/10.3390/en16010359
- Kumar A, Bhattacharjee G, Kulkarni BD, Kumar R. Role of surfactants in promoting gas hydrate formation, Industrial & Engineering Chemistry Research; 2015 Dec 16; 54(49):12217-32. https://doi.org/10.1021/acs.iecr.5b03476
- Bavoh CB, Nashed O, Khan MS, Partoon B, Lal B, Sharif AM. The impact of amino acids on methane hydrate phase boundary and formation kinetics, The Journal of Chemical Thermodynamics; 2018 Feb 1; 117:48-53. https://doi.org/10.1016/j.jct.2017.09.001
- Das S, Tadepalli KM, Roy S, Kumar R. A review of clathrate hydrate nucleation, growth and decomposition studied using molecular dynamics simulation, Journal of Molecular Liquids; 2022 Feb 15; 348:118025. https://doi.org/10.1016/j.molliq.2021.118025
- Javidani AM, Abedi-Farizhendi S, Mohammadi A, Mohammadi AH, Hassan H, Pahlavanzadeh H. Experimental study and kinetic modeling of R410a hydrate formation in presence of SDS, tween 20, and graphene oxide nanosheets with application in cold storage, Journal of Molecular Liquids; 2020 Apr 15; 304:112665. https://doi.org/10.1016/j.molliq.2020.112665
- Mohammadi A, Babakhanpour N, Javidani AM, Ahmadi G. Corn’s dextrin, a novel environmentally friendly promoter of methane hydrate formation, Journal of Molecular Liquids; 2021 Aug 15; 336:116855. https://doi.org/10.1016/j.molliq.2021.116855
- Bozorgian A, Aboosadi ZA, Mohammadi A, Honarvar B, Azimi A. Prediction of gas hydrate formation in industries, Progress in Chemical and Biochemical Research; 2020; 3: 31. 10.33945/SAMI/PCBR.2020.1.4
- Fowler DL, Loebenstein WV, Pall DB, Kraus CA. Some unusual hydrates of quaternary ammonium salts, Journal of the American chemical society; 1940 May; 62(5):1140-2. https://doi.org/10.1021/ja01862a039
- Shi and D.J.T.J.o.C.T. Liang, Semiclathrate hydrate phase behaviour and structure for CH4 in the presence of tetrabutylammonium fluoride (TBAF). 135 (2019) 252-259.
- Muromachi S, Takeya S, Alavi S, Ripmeester JA. Structural CO 2 capture preference of semiclathrate hydrate formed with tetra-n-butylammonium chloride; Cryst Eng Comm; 2022; 24(24):4366-71. https://doi.org/10.1039/D2CE00598K
- Cheng Z, Sun L, Liu Y, Xu H, Jiang L, Wang L, Song Y. Multiscale analysis of the effect of the structural transformation of TBAB semi-clathrate hydrate on CO2 capture efficiency, Energy; 2023 Jun 15:128141. https://doi.org/10.1016/j.energy.2023.128141
- Li XY, Yan J, Zhong DL, Lu SJ, Ge BB. Investigation of Tetra-n-Butyl Ammonium Bromide Semiclathrate Hydrate-Based CO2 Capture by Kinetic and In Situ Raman Spectroscopy Measurement, Industrial & Engineering Chemistry Research; 2023 Jan 25; 62(6):2504-15. https://doi.org/10.1021/acs.iecr.2c03609
- Mohammadi A, Jodat A. Investigation of the kinetics of TBAB+ carbon dioxide semiclathrate hydrate in presence of tween 80 as a cold storage material, Journal of Molecular Liquids; 2019 Nov 1; 293:111433. https://doi.org/10.1016/j.molliq.2019.111433
- Yin Z, Zheng J, Kim H, Seo Y, Linga P. Hydrates for cold energy storage and transport: A review; Advances in Applied Energy; 2021 May 26; 2:100022. https://doi.org/10.1016/j.adapen.2021.100022
- Hassan H, Javidani AM, Mohammadi A, Pahlavanzadeh H, Abedi-Farizhendi S, Mohammadi AH. Effects of Graphene Oxide Nanosheets and Al2O3 Nanoparticles on CO2 Uptake in Semi‐clathrate Hydrates, Chemical Engineering & Technology; 2021 Jan; 44(1):48-57. https://doi.org/10.1002/ceat.202000286
- Mohammadi, M. Pakzad, A.H. Mohammadi and A.J.P.S. Jahangiri, Kinetics of (TBAF + CO2) semi-clathrate hydrate formation in the presence and absence of SDS. 15 (2018) 375–384.
- Fukumoto A, Paricaud P, Dalmazzone D, Bouchafaa W, Ho TT, Fürst W. Modeling the dissociation conditions of carbon dioxide+ TBAB, TBAC, TBAF, and TBPB semiclathrate hydrates, Journal of chemical & engineering data; 2014 Oct 9; 59(10):3193-204. https://doi.org/10.1021/je500243k
- Arjmandi M, Chapoy A, Tohidi B. Equilibrium data of hydrogen, methane, nitrogen, carbon dioxide, and natural gas in semi-clathrate hydrates of tetrabutyl ammonium bromide, Journal of chemical & engineering data; 2007 Nov 15; 52(6):2153-8. https://doi.org/10.1021/je700144p
- Baghban A, Ahmadi MA, Pouladi B, Amanna B. Phase equilibrium modeling of semi-clathrate hydrates of seven commonly gases in the presence of TBAB ionic liquid promoter based on a low parameter connectionist technique, The Journal of supercritical fluids; 2015 Jun 1; 101:184-92. https://doi.org/10.1016/j.supflu.2015.03.004
- Oyama H, Shimada W, Ebinuma T, Kamata Y, Takeya S, Uchida T, Nagao J, Narita H. Phase diagram, latent heat, and specific heat of TBAB semiclathrate hydrate crystals, Fluid Phase Equilibria; 2005 Jul 28; 234(1-2):131-5. https://doi.org/10.1016/j.fluid.2005.06.005
- Sakamoto J, Hashimoto S, Tsuda T, Sugahara T, Inoue Y, Ohgaki K. Thermodynamic and Raman spectroscopic studies on hydrogen+ tetra-n-butyl ammonium fluoride semi-clathrate hydrates, Chemical engineering science; 2008 Dec 1; 63(24):5789-94. https://doi.org/10.1016/j.ces.2008.08.026
- Shimada J, Yamada M, Tani A, Sugahara T, Tsunashima K, Tsuchida Y, Hirai T. Thermodynamic Properties of Tetra-n-butylphosphonium Dicarboxylate Semiclathrate Hydrates, Journal of Chemical & Engineering Data; 2021 Dec 9; 67(1):67-73. https://doi.org/10.1021/acs.jced.1c00741
- Shimada J, Shimada M, Sugahara T, Tsunashima K, Tani A, Tsuchida Y, Matsumiya M. Phase equilibrium relations of semiclathrate hydrates based on tetra-n-butylphosphonium formate, acetate, and lactate, Journal of Chemical & Engineering Data; 2018 Aug 15; 63(9):3615-20. https://doi.org/10.1021/acs.jced.8b00481
- Zang X, Wang J, He Y, Zhou X, Liang D. Formation kinetics and microscopic characteristics of synthesized ternary gas mixture hydrates in TBAB aqueous solutions, Energy; 2022 Apr 15; 245:123327. https://doi.org/10.1016/j.energy.2022.123327
- Shi L, Shen X, Ding J, Liang D. Experimental study on the formation kinetics of methane hydrates in the presence of tetrabutylammonium bromide, Energy & Fuels; 2017 Aug 17; 31(8):8540-7. https://doi.org/10.1021/acs.energyfuels.7b01213
- Zhang F, Wang X, Lou X, Lipiński W. Effects of Tween 80 on clathrate and semiclathrate CO2 hydrate formation kinetics for carbon capture from CO2-rich gas mixtures, Carbon Capture Science & Technology; 2022 Sep 1; 4:100053. https://doi.org/10.1016/j.ccst.2022.100053
- Peng DY, Robinson DB. A new two-constant equation of state, Industrial & Engineering Chemistry Fundamentals; 1976 Feb; 15(1):59-64. https://doi.org/10.1021/i160057a011
- Javidani AM, Abedi-Farizhendi S, Mohammadi A, Mohammadi AH, Hassan H, Pahlavanzadeh H. Experimental study and kinetic modeling of R410a hydrate formation in presence of SDS, tween 20, and graphene oxide nanosheets with application in cold storage, Journal of Molecular Liquids; 2020 Apr 15; 304:112665. https://doi.org/10.1016/j.molliq.2020.112665
- Mohammadi A, Manteghian M, Mohammadi AH. Phase equilibria of semiclathrate hydrates for methane+ tetra n-butylammonium chloride (TBAC), carbon dioxide+ TBAC, and nitrogen+ TBAC aqueous solution systems, Fluid Phase Equilibria; 2014 Nov 15; 381:102-7. https://doi.org/10.1016/j.fluid.2014.08.012
- Pourranjbar M, Pahlavanzadeh H, Askari Zadeh Mahani A, Mohammadi AH. Hydrate phase equilibria of methane+ TBAC+ water system in the presence and absence of NaCl and/or MgCl2, Journal of Chemical & Engineering Data; 2020 Aug 14; 65(9):4684-91. https://doi.org/10.1021/acs.jced.0c00552
- Momeni K, Jomekian A, Bazooyar B. Semi-clathrate hydrate phase equilibria of carbon dioxide in presence of tetra-n-butyl-ammonium chloride (TBAC): Experimental measurements and thermodynamic modeling, Fluid Phase Equilibria; 2020 Mar 15; 508:112445. https://doi.org/10.1016/j.fluid.2019.112445
- Muromachi S. Phase equilibrium data for semiclathrate hydrates formed with tetra-n-butylammonium (bromide or chloride) and tetra-n-butylphosphonium (bromide or chloride) under hydrogen+ carbon dioxide pressure, Fluid Phase Equilibria; 2020 Feb 15; 506:112389. https://doi.org/10.1016/j.fluid.2019.112389
- Bollineni C, Daraboina N. Phase equilibria of methane/TBAC mixed hydrates in the presence of produced water, The Canadian Journal of Chemical Engineering; 2023 Feb; 101(2):726-34. https://doi.org/10.1002/cjce.24579
|
HOW TO CITE THIS ARTICLE Abolfazl Mohammadi. The Adverse Effect of SDS on the Growth Rate of Double Tetra n-butylammonium Chloride + Methane Semiclathrate Hydrate. Prog. Chem. Biochem. Res., 6(4) (2023) 314-326. |


